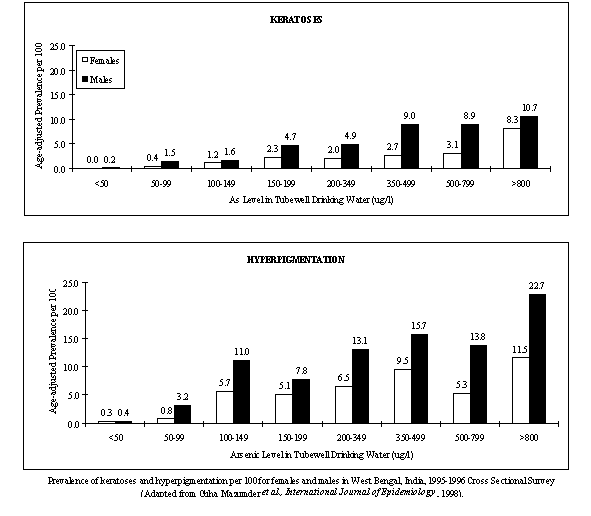
A dose-response and susceptibility investigation of skin keratoses and hyperpigmentation caused by arsenic in drinking waterAllan H. Smith1, Reina Haque1, D.N. Guha Mazumder2, Binay K. De2, Nilima Ghosh2, Soma Mitra2, David Kalman3 1. University of California, Berkeley 2. Institute of Post Graduate Medical Education and Research, Calcutta 3. University of Washington, SeattleBackground, project goals and objectivesKeratoses and hyperpigmentation are hallmark dermal signs of arsenic toxicity. Keratoses are hard raised lesions that appear on the palms and soles. Hyperpigmentation is marked by raindrop shaped diffuse dark spots on the trunk and limbs. The first detailed assessment of the dose-response relationship of arsenic induced keratoses and hyperpigmentation is underway by our group, and a key objective is to determine if susceptibility varies by arsenic methylation capability and nutritional factors, such as methionine and cysteine. Arsenic methylation will be assessed by urinary assays. Nutritional status will be determined by blood measurements of key macronutrients and micronutrients as well as by analysis of a dietary questionnaire. ApproachA case-control study has commenced which takes advantage of the largest population-based survey conducted in an arsenic-affected area of West Bengal, India. The landmark cross-sectional survey, conducted between 1995 and 1996, included over 7000 participants, 400 of whom were found to have arsenic-induced skin lesions. Approximately 280 of the 400 individuals with skin lesions were exposed to drinking water containing less than 500 *g/l of inorganic arsenic. These 280 individuals comprise the case group for our present investigation. The control group consists of lesion-free individuals randomly selected from the cross-sectional survey database, and matched on age and sex. Data obtained from personal interviews and chemical analyses of drinking water samples will be used to assess arsenic exposure. The interviews consist of questions about lifetime residential history, water sources at work, and fluid consumption. The clinical exam involves various dermatologic, neurologic, respiratory, and hepatic endpoints. A dietary questionnaire supplemented with results of blood assays will be used to ascertain the participants' nutritional status. Urinary assays will be used to determine arsenic methylation efficiency. Preliminary findingsThe interviews and sample collection commenced June 1998. Our field team reached its target rate of 5 interviews per week. To date, a little over a 100 participants have been seen. Approximately 50 urine and blood samples have been transported to the U.S. for analyses. Significance of findings By building upon an existing study, the present investigation was specifically designed to identify the shape of the dose-response curve for arsenic and skin lesions, and to determine where a threshold might lie. The study will also identify potential susceptibility factors, and thereby reduce the uncertainty in generalizing risk to other populations. Because keratoses and hyperpigmentation are the most common and earliest occurring endpoints of chronic ingestion, and because evidence suggests that these skin lesions are biomarkers of subsequent cancer risks, this study will significantly contribute to a fuller understanding of the long term health effects of consuming water containing low levels of arsenic. Next stepsInterviewing and sample collection will continue in India while data entry, and analyses of urine and blood samples will continue in the U.S. Approximately 400 individuals will be recruited by December 1999 (200 cases and 200 controls).
Corresponding author: Allan H. Smith, M.D., Ph.D., University of California, Berkeley, School of Public Health, 140 Warren Hall, Berkeley, CA 94720-7360, USA. |